The current enthusiasm among artist/printmakers for photo-etching techniques
and growing interest in electrolytic plate making methods is often accompanied
by a belief that they are new developments, and claims are made about
"new concepts" or inventions that are in fact revivals of methods
invented more than a century before. The creative and artistic uses of
techniques always follow their introduction as popular or industrial processes,
sometimes quite rapidly, at others with a long delay due to artists’ conservative
craft-based preoccupations and impatience or incomprehension, faced with
the rigour often required by the application of a new technology. Lithography
was not used seriously by artists for half a century after its invention
by Aloyis Senefelder in 1789 [1]. It took a long time for the artistic
community to accept photography as a ‘Fine Art’, and many artists used
it surreptitiously, or reacted creatively to it by rejecting photographic
illusion in painting [2].
 Charles Negre - painting based on calotype photograph |
The contemporary search for safer, non-toxic and environmentally acceptable processes has been largely provoked by the introduction of modern technology and chemicals, that had replaced safer processes and substances that were widely used in the nineteenth century, like egg albumen, gum arabic, gelatine, collodion, metal sulphates, electricity and sunlight. The fascinating early history of the development of photography is bound up with the contemporary search for photomechanical printing methods, and closely paralleled the development of electrolytic processes for printing [3]. Photography and electrolytic processes were used, together in some of the best photomechanical methods developed during the nineteenth century [4]. This article describes the genesis of these methods with some technical explanation of how they worked, only some of which have had a contemporary revival. Some that produced superb results at the time will seem unacceptably complicated today; others described, are ripe for exploitation now by resourceful artists. In particular "Electro-etching" originally invented in 1840, is being taken up very widely by printmakers, craftsmen and craftswomen, due to its methods being published freely on the internet [27].
The indisputable ‘inventor’ of photography, Joseph Nicéphore Niépce was, like many extraordinary men of that period, interested in a wide range of subjects, and he invented, among other things, the internal combustion engine with fuel injection, which he called the "Pyréolophore" patented in France in 1807 ! He began his photographic researches in 1816 from a background in lithography. He compensated for his lack of talent as a draughtsman. by using a camera obscura and was obsessed with the idea of being able to fix the images he obtained in it.
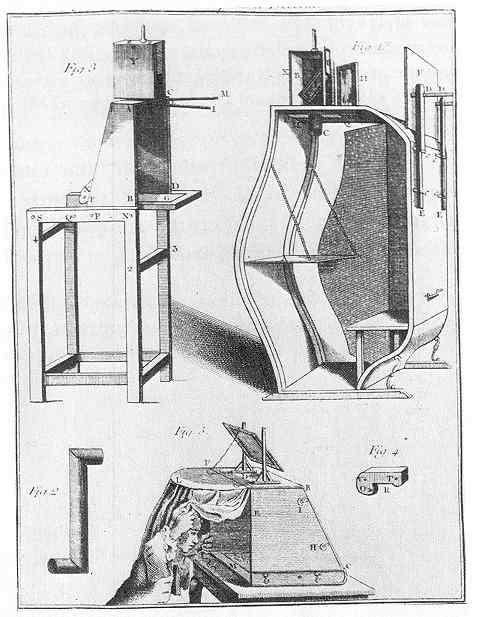 Camera Obscura types 1820 |
It was known at that time that various chemicals reacted to light and were hardened and made insoluble and so a suitable sensitized varnish would be hardened in the lightest areas [4]. This meant that if the unhardened varnish could be removed, baring the metal which could then be etched and printed in intaglio, he would have a means of simultaneously mirroring the camera obscura picture and producing a positive and permanent ink image [6]. Niépce had his first success in 1822 with bitumen of Judea mixed with oil of lavender, exposed for several hours under an engraving which was oiled to make it transparent. The areas not exposed to light could be washed away with turpentine and oil of lavender, and the dark areas etched in acid. In 1826 he produced a pewter printing plate of the Cardinal d’Amboise - the first successful attempt at photomechanical reproduction. In the same year, using his ‘heliographic’ process, he produced a photograph from nature - a view from his window - which required an exposure of eight hours in a camera obscura.
|
|
 Niepce - first photograph 1826 - view from his window - 8 hour exposure |
The theatre designer Louis Jacques Mandé Daguerre joined the race to find a way of producing a camera obscura image on an iodized silver coated copper plate. This had actually been suggested to him by Niépce in 1829. His breakthrough came in 1835 when he accidentally left an exposed plate in a closed cupboard in which a mercury thermometer had broken. It took him until 1837 to find out how to fix the image with salt, something that Henry Fox Talbot had discovered three years earlier. Niépce formed a partnership with Daguerre but died before they could publish their results [4]. His cousin Abel Niépce de St. Victor continued the partnership and invented albumen negatives. He continued work on trying to develop metal printing plates by the heliographic process.
 Daguerrotype - original version and as restored |
At that time there was an acknowledged need for an easier way of producing high quality reproductions of works of art, original works or popular views to illustrate books, which up to then required the making of steel engravings, each of which could take up to a year to produce [3, 5]. Printed photographs were perceived as the solution and the competition was later stimulated by a prize of 2000 francs offered by the Duc de Luynes in 1856 for the best method of photomechanical printing and another prize the best method for making permanent pigment-based photographic prints [3].
 Fox Talbot - photograph on salted paper 1844 |
In parallel with the development of photography, the discovery of electrolysis was exciting much interest and experimentation.. After the accidental discovery by Luigi Galvani in 1789 of ‘Galvanism’, or chemically produced electricity, Alessandro Volta built a 'galvanic battery' formed by alternating zinc and copper plates separated by fabric soaked in an acidic solution [7]. In 1834 Michael Faraday postulated his Laws of Electrolysis and established the scientific basis of the ranking of different metals according to their 'electrode potential'. Smee and Daniell invented improved versions of galvanic cells, using zinc and copper plates suspended in solutions of copper sulphates and acid separated by a permeable membrane. Mr Thomas Spencer of Liverpool made the discovery that copper was deposited on the copper plate or ‘negative metal’ and that the zinc plate was corroded or etched, and he published numerous articles on the subject in the 1840's. He and Mr John Wilson were granted a patent in 1840 for "Engraving metals by Galvanic Electricity" [9]. The process was very soon recognised and began to be used by manufacturers for marking metal, by printers for illustrative intaglio plates.
 Description of early arrangement for elecrolytic 'engraving' by Thomas Spencer 1841 |
Simultaneously Professor Jacobi of St Petersburg claimed to have made the same discovery [10]. If the cathode was a three-dimensional object or mould in wax, plaster of paris, or metal, coated with ‘plumbago’ (black lead) or graphite to conduct electricity, then the copper formed a solid negative mould over the object. The utility of this was immediately recognized and enthusiastically used to reproduce small seals and other objects by the process that became known as ‘electrotyping’ or 'galvanoplasty' [8]. Later the process was elaborated to produce much larger objects by applying a direct current from a galvanic cell to a separate cell containing a couple of metal plates in a metallic salt solution, which dissolved metal from the anode (+ve) and deposited metal on the cathode (-ve). The process of electrotyping become very widely used for creating printing plates, plating metal objects, gilding objects, decorating silverware and steel cutlery [9].
 Charles V Walker - title page of 1855 book on Electro Etching |
From then on there was a rapid development of electrolytic processes and in 1852, Charles V Walker was able to document and describe all the electrolytic processes that were currently known in his book Electrotype Manipulation, in two parts, which went through 29 editions by 1859 and was also published in the USA [11]. Part II covered those processes which were of particular application in the Arts and included detailed descriptions of a process called 'Electro-Etching',( which he said was patented in 1847), in which exactly the same equipment was used as for electrotype, but the poles were reversed and the plate on the positive pole prepared with a smoked wax ground through which lines were scratched. A single Daniell cell was used which provided a direct electric current of about one volt, and the exposed lines "..submitted to the action of the nascent oxygen.." were effectively etched. He considered this process of very great importance for artists, and it was subsequently widely used and included in every account of new methods for etchers. This process was also adapted by the steel industry for marking cutlery and other products and is still used today [16]. Charles Walker also described a novel and dangerous process of drawing directly on a plate connected to one pole of a series of batteries with an insulated stylus connected to the other - an electric arc burns an etched line which will print in intaglio[11].
Another process Charles Walker described at length was called 'Electro-tint' or ‘galvanography’ which was attributed to Professor von Kobell. "It consists in painting on white metal with etching ground or varnish:- the several shades are obtained by the relative thickness of the layers of varnish; the whole is then plumbagoed ; and the deposite obtained on it is used as a plate to furnish prints. Prof von Kobell, after obtaining a plate, examines a proof; and if too faint, he makes a mould of the plate; and having obtained a deposite, which will be similar to the original painted plate, he puts varnish on the parts which give impressions too pale, and obtains a second deposite on this, which when removed will give prints of a better character..." [11]. By 'white metal' he means a silvered copper plate and the 'deposite' is an electrotype, from which an intaglio print could be made. The word galvanography was from then on loosely used as an alternative to electrotype for specifically graphic applications. The term Galvanography was also used to describe the Jacquemin process, in which a drawing was made on a plate with lithographic ink dissolved in albumen and water. It was then heated to coagulate the albumin and make the ink insoluble, and then the plate was electrolytically etched.
After the publication of the heliographic and daguerreotype techniques in 1839, there was frantic international competition to find ways of making permanent ink prints of photographs, partly stimulated by the prize offered by the Duc de Luynes. Many of the methods used electrolytic processes in one way or another, starting off with a daguerreotype, in which the image consisted of tiny dots of mercury amalgam over a silver substrate on copper plates,. The dots acted as a slight resist to certain mordants, which would attack the silver in between. The earliest attempts, by Alfred Donne in 1839, and Joseph Berres of Vienna in 1840, simply etched the daguerreotype directly from which only a limited number of good prints could be pulled. Berres used a solid silver plate for the daguerreotype which could be etched more deeply than a silver coated copper plate, and so got more prints.
 Hippolyte Louis Fizeau - first attempt 1843 |
Hippolyte Louis Fizeau developed probably the most successful method, patenting it in 1843 [13]. He boiled the daguerreotype in potassium hydroxide to strengthen the resist dots, lightly etched it in nitric acid and then wiped it with heavy linseed oil, as if for printing in intaglio. Then he electroplated it with gold, which was deposited only on the highlights not protected by oil. He removed the oil and etched it again to deepen the dark areas, and finally electroplated the whole plate with copper to strengthen it so that many prints could be pulled [4]. The plates required some hand retouching, and the results were impressive despite difficulties in achieving good half-tones, but the method was so complicated and expensive that it never caught on.
|
Hippolyte Louis Fizeau - 1840's "Excursions Daguerriennes" |
One of the most talented early experimenters, was the painter and photographer Charles Negre who took up the methods originated by Niepce and his cousin, and elaborated them by introducing an electrolytic step, plating the partly developed steel plate with gold to protect the half tones, then aquatinting it and etching it in nitric acid. He received a French patent in 1856 and was a finalist in the Duc de Luynes competition [6].
|
Charles Negre - 1860's - competition test plate |
Meanwhile Fox Talbot had been experimenting with methods of fixing what he called ‘photogenic’ images on paper and only later joined the race to make printing plates. He patented a process called ‘photoglyphy’ in 1858, which used gelatine sensitized with potassium dichromate on a copper plate, aquatinted with gum copal powder melted onto the plate, and then etched in ferric chloride [6]. This process was later elaborated by Karl Klic of Vienna in 1879 and is now known as photogravure or heliogravure, which eventually became the most successful and widely used photo-engraving method until the introduction of new chemicals and photoresists in the 20th century. It was the noxious nature of these processes which stimulated a return to old gelatine-based methods in the 1980's and a revival of heliogravure.
|
|
 Stieglitz - photogravure 1894 |





